GD3 offers an established validated model of Passive Heymann Nephritis
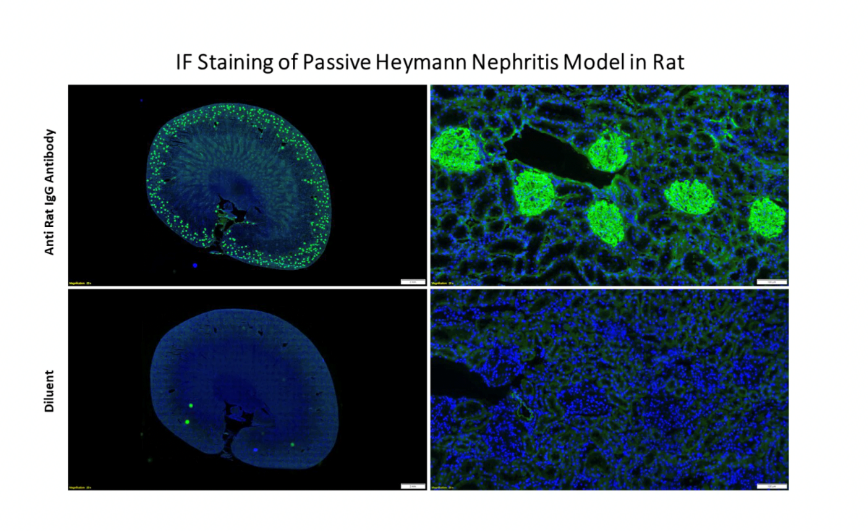
Passive Heymann nephritis (PHN) in rats resembles membranous nephropathy in humans. It is induced by injecting sheep antibody to rat proximal tubular epithelial cell brush border antigen (anti-Fx1A).
Heymann Nephritis Immune-mediated FX-1-induced glomerular nephritis is an established, robust, reproducible model at GD3.
Key Features of our established and validated anti-Fx1A-Induced PHN model for studying glomerular diseases and evaluating therapeutic interventions:
-
Accurate Disease Replication: Our meticulously developed rat model faithfully replicates the histopathological changes observed in HN. This fidelity enables precise evaluation of disease progression and
identification of
novel therapeutic targets.
-
Comprehensive Histopathological Assessment: Our expert team employs cutting-edge histopathological techniques to analyze renal tissues, providing detailed insights into the specific cellular and tissue
alterations characteristic of PHN and the disease’s pathogenesis.
-
Unveiling Mechanisms of Injury: With our rat model of PHN, you can explore the mechanisms underlying renal injury. Gain valuable insights into inflammatory responses, immune cell infiltration, glomerular
and
tubular damage, and other key pathological features, advancing your understanding of the disease and its therapeutic efficacy.
-
Reliable and Reproducible Results: Our model employs standardized protocols, ensuring consistent and reproducible results. Minimize variability and optimize preclinical research outcomes, allowing for
confident decision-making and streamlined drug development.
-
Study Design: Tailor your experiments to align with your research goals and explore the specific PHN aspects most relevant to your work. Our flexible model accommodates a range of study designs,
empowering
you to unlock critical insights into this complex disease.
-
Expert Support and Collaboration: We believe in fostering scientific collaboration and are committed to supporting your research journey. Our experienced team of scientists is dedicated to providing
guidance
and customized solutions throughout your project, ensuring your success in tackling immune-mediated renal diseases
-
Ethical Standards and Regulatory Compliance: We prioritize the welfare of animals and strictly adhere to ethical guidelines and regulatory requirements. All procedures are conducted with the utmost care
and
consideration for animal welfare, ensuring the credibility and integrity of your research.
Click here to learn more about GD3‘s fully integrated histopathology studies and cell-based services.



